Biologics Formulation Development
Stable Biologics has the capabilities to support biologics development at any stage utilizing Design of Experiment (DOE) formulation method, coupled with High-Throughput Screening (HTS) Technology to develop formulations from simple to complex formulation, starting with selection of buffers, individual excipients, to formulations consisting of a buffer and several excipients. Our laboratory possesses sophisticated instruments and equipment to develop analytical methods, perform quality controls, and stability testing.
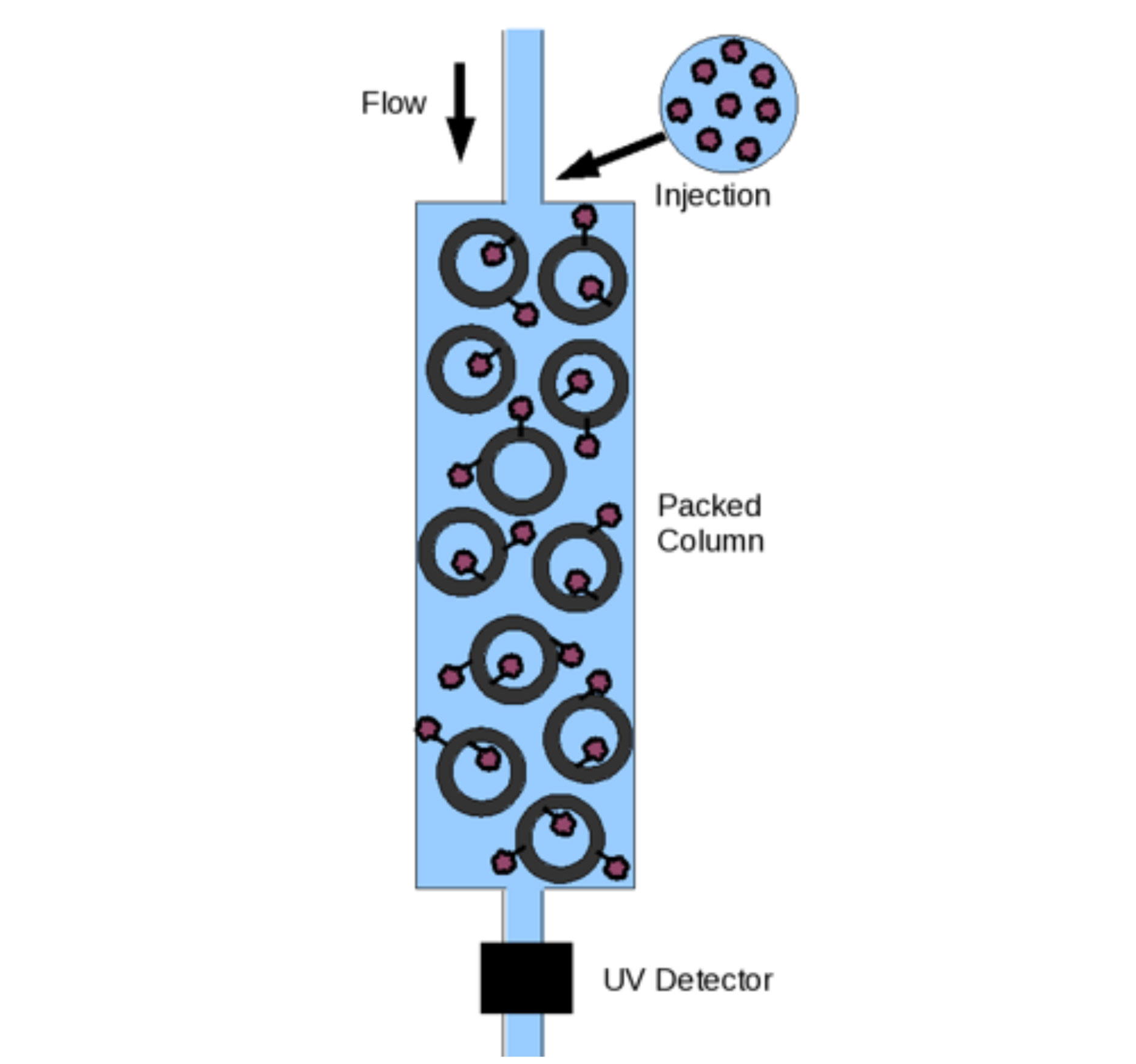
High-Throughput Formulation Screening based on Self-Interaction Chromatography Platform
Our formulation technology is based on the determination of the second virial coefficient, B22 value, a thermodynamic property of protein-protein interaction in a dilute solution via self-interaction chromatography method. High B22 value indicates that the formulation condition promotes protein solubility, while low B22 value means that the protein is unstable and has the propensity to aggregate in the solution. B22 values are typically measured by Static Light Scattering (SLS) method. However, SLS method is very time consuming and requires a substantiate quantity of protein.
Stable Biologics laboratory designed an automated capillary self-interaction chromatography system such that it requires a very small amount of protein, as low as 1 µg, to perform one formulation screen. As a result, we can screen as many as 40 conditions or excipients in 2-4 week and only requires 20-30 mg of total protein.

Method Development and Quality Controls
Our analytical team provides method development support for your drug product at any stage.
- Physical assays (aggregate and particle characterization)
- Functional assay (ELISA, qPCR, TCID50, activity, binding)
- Quality control analysis
- Impurity/degradant identification and characterization
- Higher order structure and chemical changes
- Endotoxin and microbial contaminants quantification
Stability Testing
Stable Biologics provides stability storage and/or testing services to support your drug formulation development following ICH guidelines. Our facility has temperature/humidity-controlled chambers and photostability chambers. Each chamber is equipped with online alarm notifications.
- Temperature: -80°C to +60°C
- Humidity: 25% to 90%
- Freeze thaw cycles
- Forced degradation: mechanical, chemical, photostability
Pre-Clinical Trial Support
We offer manual filling solutions for small batches and products up to biosafety level 2.

Have a formulation challenge or need analytical support?
We’re ready to collaborate.
Founded in 2008 through an academic research collaboration, we provide high-throughput biologics formulation services backed by proven science. Using advanced self-interaction chromatography (SIC), we rapidly screen protein stability with minimal material, delivering results in weeks—not months. With 30+ commercial projects completed, we offer end-to-end support from protein expression and formulation through analytical testing, stability studies, and Phase I manufacturing.